|
10/26/2019 Atoms To Mass In Grams Converter Cups
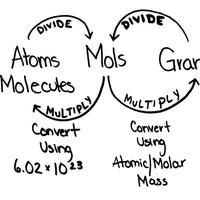
Convert grams to moles and moles to grams.Converting grams to moles is a problem that occurs when a measured amount of mass is known but the ratio for balanced reactions is needed. These two example problems show the best way to convert grams to moles and moles to grams of a molecule. Grams to Moles Example ProblemQuestion: How many moles are in 110.0 grams of CO 2.First, we need to know how many grams are in 1 mole of CO 2. This is done by finding the molecular mass of CO 2. CO 2 has 1 carbon atom and 2 oxygen atoms.
Looking at the, we can find the atomic mass of both carbon and oxygen.atomic mass of carbon = 12.0 g/molatomic mass of oxygen = 16.0 g/molmolecular mass of CO 2 = 1⋅(atomic mass of C) + 2⋅(atomic mass of O)molecular mass of CO 2 = 1⋅(12.0 g/mol) + 2⋅(16.0 g/mol)molecular mass of CO 2 = 12.0 g/mol + 32.0 g/molmolecular mass of CO 2 = 44.0 g/molThis means there are 44.0 grams in one mole of CO 2. Use this value as a conversion factor to find the number of moles of CO 2 in our problem.This setup of the conversion factor cancels out the grams, leaving the mole unit we need.# moles CO 2 = 2.5 molesAnswer: There are 2.5 moles of CO 2 in 110.0 grams of CO 2.
Moles to Grams Example ProblemQuestion: What is the mass in grams of 4.80 moles of hydrogen peroxide (H 2O 2)?Just like the first example, we need to know the molecular mass of hydrogen peroxide. H 2O 2 has two hydrogen atoms and two oxygen atoms.atomic mass of H = 1.01 g/molatomic mass of O = 16.00 g/molmolecular mass of H 2O 2 = 2⋅(atomic mass of H) + 2⋅(atomic mass of O)molecular mass of H 2O 2 = 2⋅(1.01 g/mol) + 2⋅(16.00 g/mol)molecular mass of H 2O 2 = 2.02 g/mol + 32.00 g/molmolecular mass of H 2O 2 = 34.02 g/molNow we know 1 mole of H 2O 2 has a mass of 34.02 grams. This value gives us our conversion factor to find the mass of the H 2O 2.mass of H 2O 2 = 163.30 grams4.80 moles of hydrogen peroxide has a mass of 163.30 grams. SummaryTo convert grams to moles or moles to grams:.
 Atoms To Mass In Grams Converter Cups To Cup
Find the molecular mass of the molecule. Use this value as a conversion factor. Multiply the amount by the conversion factor, making sure the undesired value is canceled out.The most common place an error is likely to occur is setting up the conversion factor incorrectly.
Grams To Atoms Chemistry
Check to make sure the correct unit cancels out.
Atoms To Mass In Grams Converter Cups To Grams
Refer to the periodic table of elements and find the atomic weight for the substance you are working with, rounding to the appropriate number of significant digits. In this case, carbon has an atomic weight of 12.0 atomic mass units (amu). The molar mass (in grams) of any substance is always numerically equal to its formula weight (in amu), so for carbon, there are 12.0 grams (g) in one mole of carbon. Write this as a fraction to the right of Step 2, again multiplying. Also, put an equal sign to the far right. It should look like this.
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
